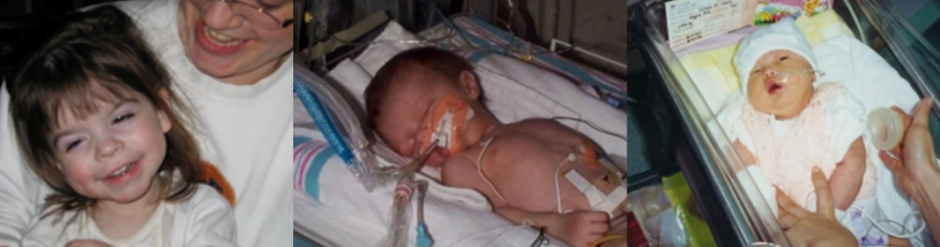
DECODE: From Subjective Assessments to Real Time Intelligence
Legacy:
Moving beyond infrequent, subjective GMFCS snapshots toward real time data. Not only mapping the brain, but providing real-time Intelligence of brain function as well as micro-improvements in kinematics.
The BRIGHT NeuroLoop CORE™ (Cognitive Orchestration & Response Engine) and EchoIntent:
- CORE™: This is the “Brain” of the operation. It handles the DTI/LNM, the AI Twin, and the closed-loop feedback.
- EchoIntent™: This is the “Heart.” It’s the user-facing interface that parents and peers interact with. It takes the complex math of the CORE and turns it into a social win.
BRIGHT’s NeuroLoop Protocol moves beyond static mapping to intelligence by creating a closed-loop neural orchestrator called the CORE™(Cognitive Orchestration & Response Engine). By integrating DTI and connectome data with real-time feedback from Magneto-Electric Nano Particles (MENPs), wearable sensors, hematological indicators and biomarkers, we create a “Living AI Twin”—a predictive model that breathes with the patient. The core of this system is its ability to distinguish between noise and statistically significant micro-improvements in motor patterns. By detecting these subtle neurological shifts before they are visible to the naked eye, the AI determines the trajectory of recovery in real-time. This isn’t just data collection; it is active intelligence. When the system detects a positive trend, it reinforces the current neural pathway, locking in the progress. If the data suggests a plateau or regression, the AI instantly pivots, redirecting the exosuit’s assistance and EMG stimulation parameters to find a more effective mechanical and electrical path. By constantly iterating between “detect, predict, and redirect,” the system transforms passive rehabilitation into a dynamic, hyper-personalized evolution of the human nervous system.
EchoIntent™ is a Verbal Intent Generator and is the NeuroLoop’s solution to the “social atrophy” caused by severe HIE, Stroke, and Brian Injury, transforming the victim from a passive recipient of care into an active social agent. By moving beyond simple communication “need-based” signals like hunger or pain, this system utilizes a Multimodal AI Engine to synthesize real-time data from infrared eye-tracking, facial micro-expressions, “gruntal” acoustics, and body language to infer complex social intent and convert that into real-time voice generation. This inferred intent is then cross-referenced with RFID-enabled room sensing and a deep-memory database of the child’s/victim’s interests in order to create context based suggested “conversation starters”.
The Frontier Edge:
The objective is not merely to identify the lesion, but to determine which neural circuits remain viable for activation and provide real-time closed-loop feedback. This provides the precise coordinates for the PRIME phase.
AI Digital Twins Simulate Years of Clinical Trails in Seconds
Technical Glossary:
- CORE™ (Cognitive Orchestration & Response Engine): BRIGHT’s closed-loop neural orchestrator integrating DTI and connectome data with real-time feedback from Magneto-Electric Nano Particles (MENPs), wearable sensors, hematological indicators and biomarkers, creating a “Living AI Twin”.
- EchoIntent™: BRGHT’s Verbal Intent Generator that utilizes a Multimodal AI Engine to synthesize real-time data from infrared eye-tracking, facial micro-expressions, “gruntal” acoustics, and body language to infer complex social intent and convert that into real-time voice generation.
- DTI (Diffusion Tensor Imaging): An advanced MRI technique that maps the diffusion of water molecules to visualize the white matter tractography of the brain.
- Connectome Mapping: The comprehensive mapping of neural connections in the brain to understand functional and structural organization.
- MENPs (Magneto-Electric Nano Particles): Metallic nanoparticles acting as wireless transducers, converting internal brain electric fields into magnetic signals for high-resolution, real-time neuro-rehabilitation monitoring.
- AI Digital Twin: A virtual model of a patient’s neurological architecture used to simulate treatment outcomes and recovery trajectories. Use real-time data from wearables, infrared, video, and biomarkers. Predict movement intention in real time and access plasticity changes as they happen. Adjust and improve in real time.
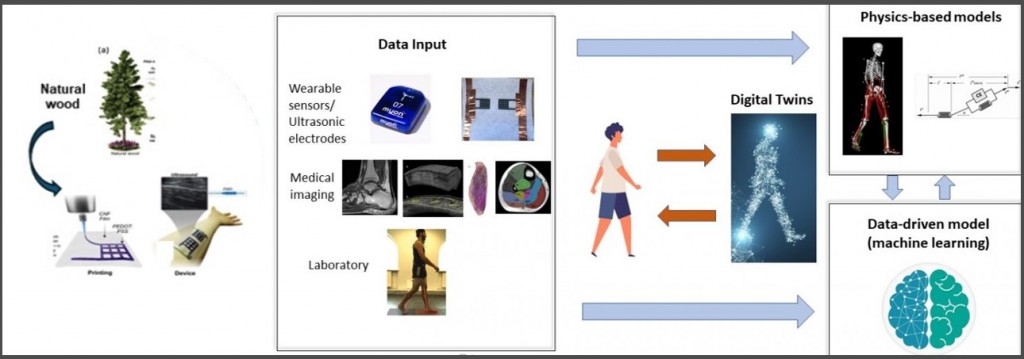
Photo Credit: https://www.digitalfutures.kth.se/profile/yuanyuan-li/
Scholarly Citations:
ETH Zurich (SCAI Lab): The Spinal Cord Injury & Artificial Intelligence (SCAI) Lab specializes in multimodal sensing to create digital twins for long-term recovery monitoring.
- Lauer-Schmaltz, M. W., et al. (2024). “Human Digital Twins in Rehabilitation: A Case Study on Exoskeleton and Serious-Game-Based Stroke Rehabilitation Using the ETHICA Methodology.” IEEE Access.
- Significance: This is a foundational study using the ETHICA methodology to adjust exoskeleton assistance and game difficulty in real-time, predicting muscle fatigue with high accuracy to optimize upper-limb recovery.
Most Recent (Post-Stroke Aphasia):
- Author(s) Unlisted (Feb 2026). “Digital Twin Model of Treatment Outcomes in Post-Stroke Aphasia.” PMC.
- Significance: This recent study applies a digital twin framework to chronic post-stroke aphasia, successfully predicting over 58% of the variance in naming performance during language treatment.
Most Impactful (HIE & Neonatal Brain Injury):
- Author(s) Unlisted (Aug 2025). “Digital biomarkers as predictors of brain injury in neonatal hypoxic-ischemic encephalopathy.” PMC.
- Significance: This study uses real-time bedside clinical data (heart rate, BP, oxygenation) during therapeutic hypothermia to stratify brain injury severity in neonates, serving as a predictive “neuro-twin” for ICU decision-making.
Most Recent (Critical Care Modeling):
- Gao, Y. et al. (2025). “Toward Digital Twins in the Intensive Care Unit: A Medication Management Case Study.” PMC.
- Significance: While broadly for the ICU, this framework is being applied to the Neurocritical Care (NCC) environment at Anschutz to simulate patient responses to complex drug regimens following acute brain insults
Additional:
- Bax, M., et al. (2006). “The definition and classification of cerebral palsy.” Developmental Medicine & Child Neurology.
- Panigrahy, A., et al. (2009). “Relationship of White Matter Tractography to Function in Children with CP.” NeuroImage.
- Van den Heuvel, M. P., & Sporns, O. (2011). “Rich-club organization of the human connectome.” Journal of Neuroscience.
Launch into The NeuroLoop:
MAP | CLEAR | UNLOCK | UPDATE | FUEL | SYNC | TUNE | PRIME | GUIDE | CEMENT